Lyons, Suzi
(2013)
Suboxone feasibility study evaluated.
Drugnet Ireland,
Issue 47, Autumn 2013,
pp. 19-22.
In June 2013 the Department of Health released the results of an evaluation of the Suboxone feasibility study which started in June 2009.1 The evaluation was carried out between October 2010 and February 2011. The objectives of the evaluation were to:
· evaluate how patients were selected for participation and how they progressed through the study;
· examine how the prescribing and dispensing of Suboxone operated in an Irish context;
· consider the practical operation of the feasibility study;
· identify the core elements of the regulatory framework needed for the safe use of Suboxone in Ireland.
A variety of methods was used to evaluate the feasibility study: analysis of quantitative and clinical records, semi-structured interviews, and surveys. Not all patients who had started on Suboxone could be contacted for consent to be included in the evaluation and not all patient records were available to be audited. Other stakeholder involvement consisted of: eight semi-structured interviews with prescribers; 13 semi-structured telephone interviews with pharmacists/dispensers; 36 responses to semi-structured survey of patients (completed either on-line, on paper or over the telephone); two face-to-face interviews with patients and two written submissions from patients. The clinical notes and dispensing records were reviewed for 41 patients. There were five key informant interviews.
A total of 139 patients registered as having received Suboxone between 2006 and 2011 were eligible to be included in the evaluation. Also included were 11 prescribers based in the Drug Treatment Centre Board (DTCB), seven prescribing GPs based in other clinics or practices, and 50 dispensers, the majority (78%) of which were community pharmacies. Table 1 outlines the characteristics of those included in the evaluation and the reason for exit.
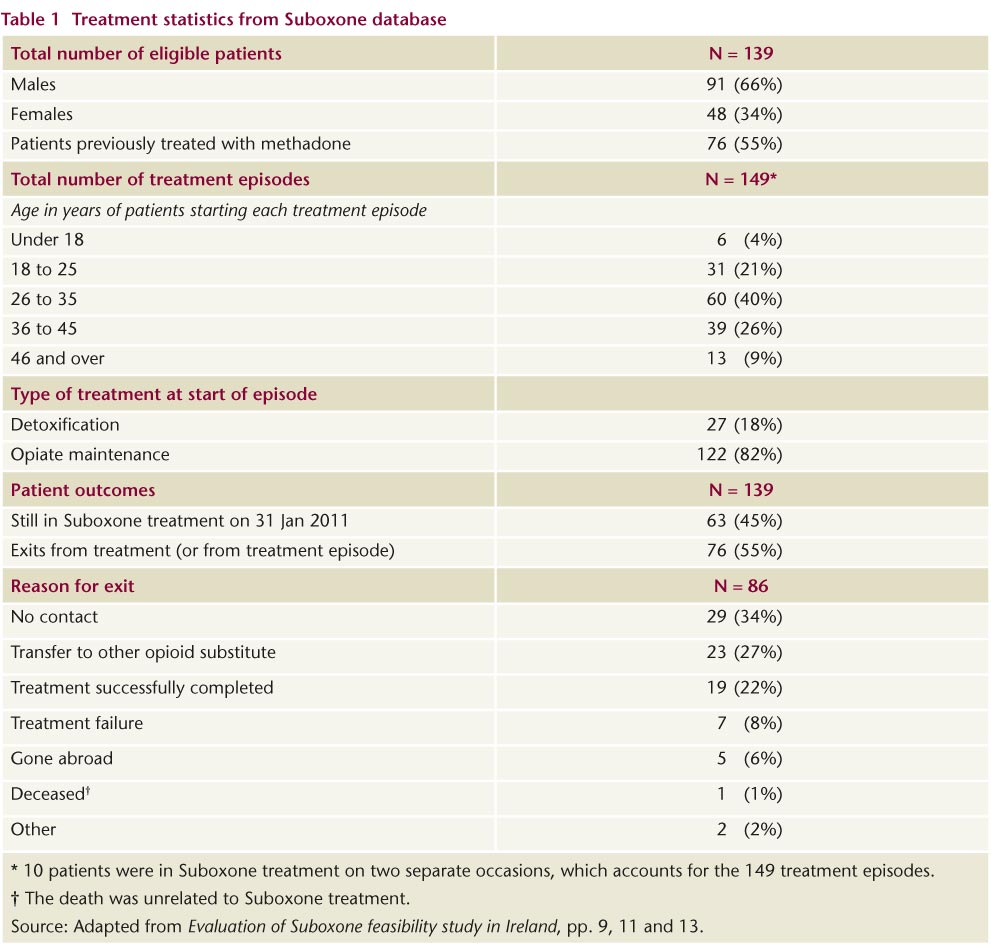
* 10 patients were in Suboxone treatment on two separate occasions, which accounts for the 149 treatment episodes.
† The death was unrelated to Suboxone treatment.
Source: Adapted from Evaluation of Suboxone feasibility study in Ireland, pp. 9, 11 and 13.
Process and limitations of the study
The author noted several issues that may influence the results and generalisability of the evaluation. She was unable to contact a number of patients who had received Suboxone but who were no longer in contact with the treatment services. Not all who consented to be included in the evaluation could be contacted subsequently. Also, the evaluation only included the opinions of those who had been involved in the original feasibility study,, who therefore might already have been positively disposed to the use of Suboxone.
Rationale for commencing Suboxone
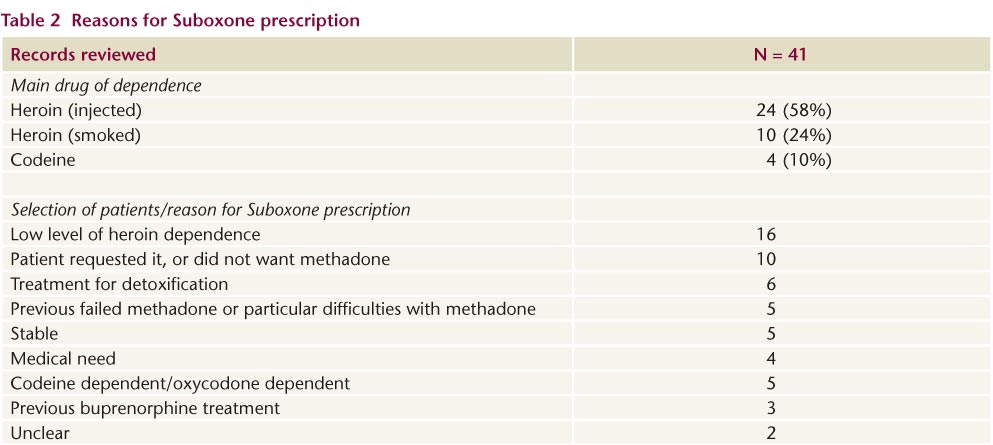
The main reasons for prescribing Suboxone, as recorded in the clinical notes or dispensing record for 41 patients, were: patient had low level of heroin dependence, either having never injected had rarely injected or had a short history of heroin use (16, 39%), and patient requested it, or did not want methadone (10, 24%) (Table 2). The survey of patients showed that the majority (79%) felt that Suboxone was the best option for them after discussions with their doctor. Of 22 patients previously prescribed methadone, 45% had experienced side-effects. Of 11 who had never been prescribed methadone, 55% wanted detoxification (i.e. to be substance free) rather than to stay on opiate substitution treatment.
Suboxone tablets caused some difficulties for pharmacists. Patients were supplied with a variety of information documents and/or verbal advice on starting Suboxone. However some of the patients reported that they had forgotten what they had been told when first prescribed Suboxone, highlighting the need for standardised, accessible written information.
Participants were asked about the possibility of diversion of Suboxone. The consensus was that there was little or no diversion due to the tight controls and stability of the patients. In the patient survey, 76% reported that Suboxone was not available on the street.
Costs
This evaluation discusses the 2007 report done by the National Centre for Pharmacoeconomics (NCPE) for the expert group.2 That report concluded that on current evidence Suboxone could not be considered cost effective for patients attending HSE clinics unless opiate abstention rates were at least 10% higher with Suboxone than with methadone.The evaluation noted that the low cost of methadone makes comparing costs with any other treatment difficult. It was felt that societal costs had not been factored into the NCPE report and that, although more expensive than methadone, providing Suboxone to certain patients for whom there is a clear rationale would have economic and societal benefits.
Regulating provision
There was no clear consensus among prescribers or key informants as to what was the most appropriate mechanism to regulate provision, as each had both positive and negative aspects. The issues discussed were:
· having a cap on the number of patients who can be prescribed Suboxone;
· having a fixed budget cap (rather than a cap on patients);
· restricting prescribing to certain groups of prescribers and/or patients;
· negotiation with the relevant pharmaceutical company on price.
Conclusions and recommendations
The evaluation concludes that ‘Suboxone has been used in this feasibility study with a wide range of patient groups, for whom it has the potential to be beneficial and it appears to offer a number of advantages over methadone’ (p.44). In considering future safe provision of Suboxone in Ireland, it makes a number of recommendations (pp.44–47), which are summarised below.
1. The Suboxone Expert Group discuss and agree interim criteria for who should be eligible for Suboxone treatment.
2. If Suboxone prescribing is restricted to certain groups/subjected to certain criteria in future, audit and reporting processes should be established to add weight to the criteria and enable any prescribing patterns that are unusual or fall outside the criteria to be identified and explored.
3. National guidelines on the use of Suboxone across all prescribing and dispensing settings should be developed.
4. Standard patient information resources should be used across all settings.
5. Prescriptions for Suboxone should be designed for clarity and avoidance of ambiguity.
6. The system for remuneration of Suboxone in future should be incorporated into remuneration systems for opioid treatment services and should be clearly communicated to prescribers and dispensers (including any future prescribers and dispensers).
7. Clarification of the ethical and legal position of pharmacists and clinics in relation to the various options for coping with stock shortages should be provided. Professional guidance should be prepared in relation to the appropriate management of such situations.
8. Arrangements should be made for the appropriate management of Suboxone stock which may be left in pharmacies where they no longer have any patients on the drug.
9. Suboxone should be made available through more than one wholesaler to reduce the likelihood of stock shortages.
10. Suboxone initiation and prescribing should be restricted to those with suitable expertise and training.
11. Suboxone dispensing should be restricted to those with suitable expertise and training.
12. There is a need for a mechanism for prescribers to share and discuss their experiences with Suboxone (and in the management of opioid dependence in general, including methadone).
13. Engagement should take place with the pharmaceutical company marketing Suboxone in order to consider price, budget projections and shared-risk arrangements for any future use of Suboxone.
14. Any future consideration of the cost-effectiveness of Suboxone should take into account the full range of potential benefits from successful treatment of opioid dependence, including reduced social costs.

